Introduction: The Critical Role of Implant Design
Implant design is a fascinating and profoundly impactful field, sitting at the intersection of engineering, materials science, and medicine. For engineers, it’s about crafting devices that restore function, relieve pain, and improve the quality of life for patients. Whether we’re talking about orthopedic prostheses, cardiovascular stents, or dental implants, the underlying principles of robust engineering design are paramount. A well-designed implant integrates seamlessly with biological systems, withstands complex physiological loads, and ensures long-term safety and efficacy.
This guide dives deep into the engineering considerations behind successful implant design, offering practical insights for professionals in biomechanics, structural engineering, and related fields. We’ll explore material selection, geometric optimization, advanced simulation techniques, and manufacturing challenges, all geared towards creating resilient and biocompatible medical devices.

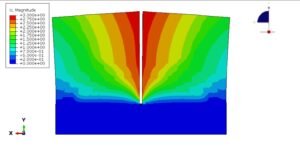
Image: Finite Element Analysis illustrating stress distribution in a dental implant and surrounding bone. Courtesy of Dr. G. R. Bafna via Wikimedia Commons.
The Pillars of Effective Implant Design
Successful implant design isn’t a single magical step; it’s a careful orchestration of several critical elements. Neglecting any one of these can lead to complications, premature failure, or suboptimal patient outcomes. Let’s break down the key considerations.
Material Selection: Beyond Biocompatibility
Choosing the right material is arguably the first and most crucial decision in implant design. It’s not just about avoiding adverse reactions; it’s about matching mechanical properties to the physiological environment and ensuring long-term durability. Engineers must consider:
- Biocompatibility: The material’s ability to perform with an appropriate host response in a specific application. It means no toxicity, no inflammation, and no allergic reactions.
- Biostability: Resistance to degradation in the highly corrosive physiological environment.
- Mechanical Properties: Strength, stiffness (elastic modulus), fatigue resistance, wear resistance, and toughness must all be tailored to the implant’s function and the loads it will experience.
- Manufacturability: Can the material be processed into the desired complex geometry economically and consistently?
- Sterilizability: Can the material withstand sterilization methods without degrading?
Common Materials in Implant Design
Here’s a quick look at some widely used materials and their typical applications:
| Material Class | Examples | Key Properties | Typical Applications |
|---|---|---|---|
| Metals | Titanium & Ti-alloys (Ti-6Al-4V), Cobalt-Chromium alloys (CoCrMo), Stainless Steel (316L) | High strength-to-weight, excellent fatigue resistance, corrosion resistance, osseointegration (Ti). | Orthopedic implants (hips, knees), dental implants, spinal fixation, cardiovascular stents. |
| Polymers | UHMWPE (Ultra-High Molecular Weight Polyethylene), PEEK (Polyether Ether Ketone), PMMA (Polymethyl Methacrylate) | Low friction, wear resistance (UHMWPE), radiolucency, tunable stiffness (PEEK), bone cement (PMMA). | Articular surfaces in joint replacements, spinal cages, drug delivery systems, bone cements. |
| Ceramics | Alumina, Zirconia, Hydroxyapatite (HA) | High hardness, wear resistance, excellent biocompatibility, bone-like properties (HA). | Dental crowns, femoral heads, coatings for metal implants (HA). |
| Composites | Carbon fiber reinforced PEEK | Tunable stiffness, high strength, radiolucency. | Spinal cages, fracture fixation plates (investigational). |
Geometric Considerations: Form Meets Function
Once a material is chosen, the geometry dictates how the implant interacts with the surrounding tissues and distributes mechanical loads. This is where CAD tools like CATIA or SolidWorks are indispensable.
- Anatomical Fit: The implant must conform accurately to the patient’s anatomy to ensure stability and proper load transfer. Custom-fit implants derived from patient-specific imaging (CT/MRI) are becoming more common, facilitated by additive manufacturing.
- Stress Shielding: A critical phenomenon where a stiff implant carries too much load, causing the surrounding bone to atrophy (resorb) due to lack of stress. Engineers aim for a stiffness match between the implant and bone, or employ porous structures to encourage load transfer.
- Surface Features: Micro and macro-level surface features are crucial for promoting biological integration. Roughened, porous, or coated surfaces can enhance osseointegration – the direct structural and functional connection between living bone and the surface of a load-bearing artificial implant.
- Minimizing Stress Concentrations: Sharp corners, abrupt changes in cross-section, and tight radii act as stress concentrators, leading to premature fatigue failure. Thoughtful radii and smooth transitions are key.
- Anchoring Mechanisms: How the implant is secured – cemented, press-fit, or screw-fixed – heavily influences its geometry and long-term stability.
Surface Topography and Osseointegration
For bone-interfacing implants, surface engineering is a science in itself. Techniques include sandblasting, acid etching, plasma spraying, and anodization to create micro-scale roughness. These features provide a scaffold for bone cells to attach and proliferate, significantly improving implant stability and longevity. Engineers often coat implants with Hydroxyapatite (HA) to mimic natural bone composition, further enhancing integration.
Loading Environment & Biomechanics: The Real-World Challenge
Implants don’t exist in a static world. They are subjected to complex, dynamic, and often unpredictable forces within the body. Understanding these biomechanical forces is vital.
- Static vs. Dynamic Loads: A hip implant experiences both static body weight and dynamic forces from walking, running, or jumping, which can be several times body weight.
- Fatigue Loading: Most implant failures are due to fatigue – repeated loading cycles over many years. Predicting fatigue life requires sophisticated analysis and experimental testing.
- Wear and Fretting: In articulating joints (like knees or hips), relative motion between components leads to wear, generating particles that can cause inflammation or osteolysis (bone loss). Fretting corrosion can occur at interfaces subject to small oscillatory movements.
- Fluid Dynamics: For cardiovascular implants like heart valves or stents, understanding blood flow patterns (shear stress, turbulence) through CFD is critical to prevent thrombosis (blood clot formation) and ensure long-term function.
Understanding In-Vivo Forces: A Design Checkpoint
Engineers often consult biomechanical databases or perform gait analysis studies to estimate typical forces on implants. For example, a hip implant might need to withstand 2-3 million cycles per year, with peak loads reaching 3-5 times body weight. These real-world loading conditions directly inform fatigue analysis and material selection criteria.
The Role of Simulation in Implant Design
Before a single prototype is built, advanced computational tools allow engineers to predict implant behavior under various conditions, significantly reducing development time and costs. This is where CAD-CAE workflows truly shine.
Finite Element Analysis (FEA) for Implants
FEA is the cornerstone of modern implant design. It allows engineers to simulate stress, strain, deformation, and even predict fatigue life and potential failure modes. Software like Abaqus, ANSYS Mechanical, and MSC Nastran are standard tools in this domain.
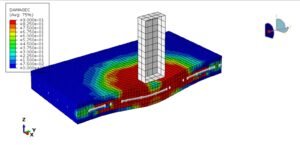
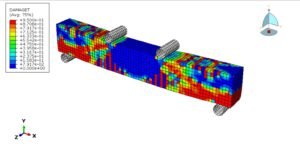
- Stress Distribution: Visualize where stresses concentrate, helping to optimize geometry and prevent stress shielding.
- Deformation Analysis: Predict how the implant and surrounding tissue deform under load, ensuring proper fit and function.
- Fatigue Life Prediction: Estimate how many loading cycles an implant can withstand before failure, crucial for long-term durability.
- Osseointegration Potential: By analyzing micromotion and strain at the bone-implant interface, FEA can provide insights into conditions favorable for bone growth.
- Failure Analysis: Simulate fracture initiation and propagation under extreme loads.
Practical FEA Workflow for Implant Design
- CAD Model Preparation: Start with a precise 3D model of the implant and often the surrounding anatomical structures (bone, cartilage, soft tissue). Simplifying complex anatomical geometries is often necessary without losing critical detail.
- Material Properties Assignment: Accurately define material properties for all components – implant, bone (often anisotropic), cartilage (hyperelastic), etc. This is crucial for realistic simulation.
- Meshing: Discretize the geometry into a finite number of elements. Fine meshes are needed in critical areas like interfaces or stress concentrators, while coarser meshes can be used elsewhere to optimize computational cost. Tools like HyperMesh or the meshing capabilities within Abaqus/ANSYS are used.
- Boundary Conditions (BCs) & Loading: Apply realistic physiological loads (e.g., muscle forces, joint reaction forces) and constraints (e.g., fixed ends of bones) that mimic the in-vivo environment. Pitfalls include over-constraining the model or applying unrealistic load magnitudes or directions.
- Solver Execution: Run the FEA solver (e.g., Abaqus Standard/Explicit, ANSYS Mechanical). Non-linear analyses (contact, large deformation, material non-linearity) are common for biomechanical problems.
- Post-Processing: Interpret results visually (stress plots, deformation animations) and numerically (stress values, strain energy). Identify critical regions, evaluate safety factors, and suggest design improvements.
Verification & Sanity Checks in FEA
Even the most advanced FEA models are only as good as their inputs and assumptions. Rigorous verification is non-negotiable.
- Mesh Convergence Study: Ensure that refining the mesh further does not significantly change the results. This confirms that the discretization error is acceptable.
- Boundary Condition Realism: Double-check that BCs accurately represent the physiological environment. Are the fixed points correct? Are the applied loads distributed realistically?
- Material Property Review: Verify that material properties (elastic modulus, Poisson’s ratio, yield strength, fatigue curves) used are appropriate for the specific tissue/material and loading conditions.
- Analytical Checks/Hand Calculations: For simplified sub-components or initial design iterations, compare FEA results with basic analytical solutions (e.g., beam theory, stress concentration factors) to catch gross errors.
- Convergence Checks: For non-linear solutions, monitor convergence plots to ensure the solution has converged robustly. Divergence can indicate instability or incorrect setup.
- Validation with Experimental Data: Whenever possible, validate simulation results against physical tests (e.g., mechanical testing of prototypes, in-vitro cadaver studies). This is the ultimate proof of model accuracy.
- Sensitivity Analysis: Evaluate how changes in input parameters (e.g., material properties within their variability range, slight variations in load) affect the output. This provides insights into design robustness.
For complex models requiring substantial computational power, EngineeringDownloads offers affordable HPC rental to run your large-scale FEA models efficiently.
Computational Fluid Dynamics (CFD)
While less common for orthopedic implants, CFD (using tools like ANSYS Fluent/CFX or OpenFOAM) is vital for designing cardiovascular devices like stents or heart valves. It allows engineers to predict blood flow patterns, wall shear stress, and potential regions of recirculation that could lead to thrombosis.
Multibody Dynamics (MBD)
Software like ADAMS is used for MBD simulations to analyze the kinematics and dynamics of joint replacements. This helps understand how components interact, predict wear, and optimize implant placement for optimal range of motion.
Manufacturing & Prototyping
The best design on paper is useless if it cannot be manufactured precisely and repeatably. Manufacturing choices profoundly impact cost, material properties, and geometric fidelity.
Additive Manufacturing (AM) for Implants
Also known as 3D printing, AM is revolutionizing implant design. Techniques like Selective Laser Melting (SLM) for metals (titanium, cobalt-chrome) or Fused Deposition Modeling (FDM) for polymers offer:
- Customization: Patient-specific implants tailored to unique anatomies.
- Complex Geometries: Fabrication of intricate porous structures to promote bone ingrowth and reduce stress shielding, which are difficult or impossible with traditional methods.
- Reduced Waste: Near-net-shape manufacturing, especially valuable for expensive biomaterials.
Traditional Manufacturing Techniques
While AM is growing, traditional methods remain crucial:
- Machining: Precise removal of material using CNC machines, ideal for achieving tight tolerances and smooth surfaces.
- Forging/Casting: Used for creating initial blanks or near-net shapes for some metal implants, offering good mechanical properties through grain refinement (forging).
- Surface Treatments: Polishing, anodizing, and coating (e.g., HA coatings) are critical post-processing steps.
Prototyping & Mechanical Testing
Before clinical use, prototypes undergo rigorous mechanical testing according to international standards (e.g., ISO 7206 for hip implants, ISO 14801 for dental implants). This includes fatigue testing, static strength tests, and wear tests to validate design assumptions and material performance.
Design Validation & Regulatory Landscape
Bringing an implant to market is a highly regulated process to ensure patient safety and efficacy.
Pre-clinical Testing: Beyond the Computer
- In-vitro Testing: Laboratory tests simulating physiological conditions (e.g., mechanical loading in bioreactors, corrosion tests).
- In-vivo Animal Studies: Used to evaluate biocompatibility, osseointegration, and long-term performance in a living system before human trials.
Regulatory Pathways: The Gateway to Patients
Understanding the regulatory requirements is crucial. In the USA, the FDA (Food and Drug Administration) oversees medical devices, while in Europe, the CE Mark is required. These bodies ensure that implants meet stringent safety and performance standards through a process that includes detailed design documentation, manufacturing controls, and extensive testing data.
Post-Market Surveillance: Continuous Improvement
Even after market approval, implant performance is continuously monitored. Post-market surveillance helps identify rare complications, assess long-term outcomes, and inform future design improvements. This feedback loop is essential for continuous engineering innovation in medical devices.
Common Pitfalls in Implant Design
Even experienced engineers can stumble. Here are some common mistakes to avoid:
- Inadequate Material Selection: Choosing a material solely based on strength, neglecting biocompatibility, fatigue resistance, or corrosion behavior.
- Poor Geometric Fit: Designs that don’t conform well to patient anatomy, leading to micromotion, instability, or soft tissue impingement.
- Neglecting Fatigue: Underestimating the number of load cycles or the magnitude of peak stresses, leading to premature fatigue failure.
- Insufficient Testing: Skipping critical mechanical tests or not adhering to relevant ISO standards.
- Ignoring Biomechanics: Failing to accurately characterize the in-vivo loading environment or the biological response to the implant.
- Over-Reliance on Simulation Alone: While powerful, simulation is a tool, not a substitute for physical testing and validation.
- Lack of Manufacturing Awareness: Designing complex features that are impossible or prohibitively expensive to manufacture to the required precision.
Leveraging Advanced Tools & Expertise
Modern implant design increasingly relies on advanced computational tools. Python and MATLAB, for instance, are invaluable for automating pre-processing (e.g., generating patient-specific meshes from medical images), post-processing FEA results, and performing statistical analysis of test data. These scripting capabilities enhance efficiency and allow for more robust design exploration.
For teams looking to upskill, EngineeringDownloads provides online/live courses and internship-style training in advanced FEA, CAD-CAE integration, and biomechanical simulation. We also offer project and contract consultancy to help overcome complex implant design challenges.
Conclusion: Engineering for a Better Tomorrow
Implant design is a testament to the power of engineering to directly improve human lives. It demands a multidisciplinary approach, blending deep understanding of materials, mechanics, biology, and advanced computational tools. By meticulously considering material properties, optimizing geometry, rigorously simulating performance, and adhering to strict validation protocols, engineers can craft devices that offer durable and effective solutions for patients worldwide.
The field continues to evolve rapidly with innovations in smart materials, bio-resorbable implants, and even more patient-specific solutions driven by AI and advanced manufacturing. For engineers in this domain, it’s a journey of continuous learning and profound impact.
Further Reading
For more detailed information on standards for surgical implants, refer to the International Organization for Standardization (ISO) technical committee on Implants for surgery: ISO/TC 150 – Implants for surgery
Frequently Asked Questions (FAQs)
Here are some common questions about implant design:
What is the primary goal of implant design?
The primary goal of implant design is to create medical devices that can replace or support biological structures, restoring function, relieving pain, and improving patient quality of life. This involves ensuring biocompatibility, mechanical durability, and optimal integration with the body.
Why is material selection so critical in implant design?
Material selection is critical because it dictates an implant’s biocompatibility (how the body reacts to it), its mechanical properties (strength, stiffness, fatigue resistance), its resistance to corrosion in the body, and its manufacturability. The wrong material can lead to rejection, premature failure, or adverse tissue reactions.
How does Finite Element Analysis (FEA) contribute to implant design?
FEA allows engineers to computationally simulate how an implant and surrounding tissues will behave under various physiological loads. It helps predict stress distribution, deformation, potential stress shielding, and fatigue life, enabling design optimization before physical prototyping and testing, thus saving time and resources.
What is stress shielding, and how do engineers address it?
Stress shielding occurs when a stiff implant carries too much load, leading to reduced stress in the adjacent bone. This can cause the bone to weaken and resorb over time (Wolff’s Law). Engineers address this by trying to match the implant’s stiffness closer to that of bone, using porous structures, or optimizing implant geometry to encourage more natural load transfer to the bone.
What are the main challenges in manufacturing medical implants?
Manufacturing medical implants presents challenges such as achieving extremely tight tolerances, working with specialized biomaterials, maintaining sterility, and producing complex geometries. Additive manufacturing (3D printing) is increasingly used to overcome some of these challenges, especially for patient-specific or intricate designs.
Why are regulatory standards important for implant design?
Regulatory standards (like those from the FDA or for CE Marking) are crucial because they ensure that medical implants meet stringent safety, quality, and performance criteria before they can be used in patients. Adherence to these standards involves extensive documentation, testing, and validation to protect public health.
What role does biomechanics play in implant design?
Biomechanics is fundamental to implant design as it involves understanding the mechanical forces and motions within the human body. Engineers apply biomechanical principles to analyze physiological loads, predict how an implant will interact with biological tissues, and design for long-term function and durability, considering aspects like fatigue, wear, and fluid dynamics.